| By K. Lee Lerner |
Citing their joint “contributions to our understanding of the evolution of the universe and Earth’s place in the cosmos,” the Royal Swedish Academy of Sciences awarded equal shares of the 2019 Nobel Prize in Physics to Canadian-born American physicist James Peebles (1935‒), the Albert Einstein Professor of Science at Princeton University in the United States, and divided the other half of the prize between two Swiss physicists at the University of Geneva, Michel Mayor (1942‒) and Didier Queloz (1966‒). Queloz also holds a professorship at the University of Cambridge in the United Kingdom.
Peebles was specifically cited “for theoretical discoveries in physical cosmology” that have helped shape the study of cosmology (the structure and origin of the universe), and our understanding of the big bang (the explosive expansion of the cosmos from a hot and dense singularity). Peebles’s work helped to correctly interpret the lingering radiation from the big bang that took place almost 14 billion years ago. Peebles also helped provide evidence that offers ongoing challenges to current and future astronomers: that only about 5 percent of the material universe is known, and that 95 percent of the universe remains unknown dark matter and dark energy.
Mayor and Queloz were cited for the “discovery of an exoplanet orbiting a solar-type star.” Their 1995 discovery of planet 51 Pegasi, made while working at the Haute-Provence Observatory in southern France, was the first evidence of a planet outside our solar system in orbit around another star in our Milky Way galaxy. Subsequent studies have shown 51 Pegasi to be largely composed of gas and similar in structure to Jupiter. The work of Mayor and Queloz, including advances they made in instrumentation, has since spurred the discovery of about 4,000 exoplanets within the Milky Way that vary in size, composition, and orbits around their stars.
“For the development of lithium-ion batteries,” the Academy equally divided the 2019 Nobel Prize in Chemistry among the German-born American engineer John B. Goodenough (1922‒), a professor of engineering at the University of Texas at Austin; British-born American scientist M. Stanley Whittingham (1941‒), a professor at Binghamton University, State University of New York; and Japanese scientist Akira Yoshino (1948‒), a professor at Meijo University, Nagoya, Japan, and honorary fellow at the Asahi Kasei Corporation in Tokyo, Japan.
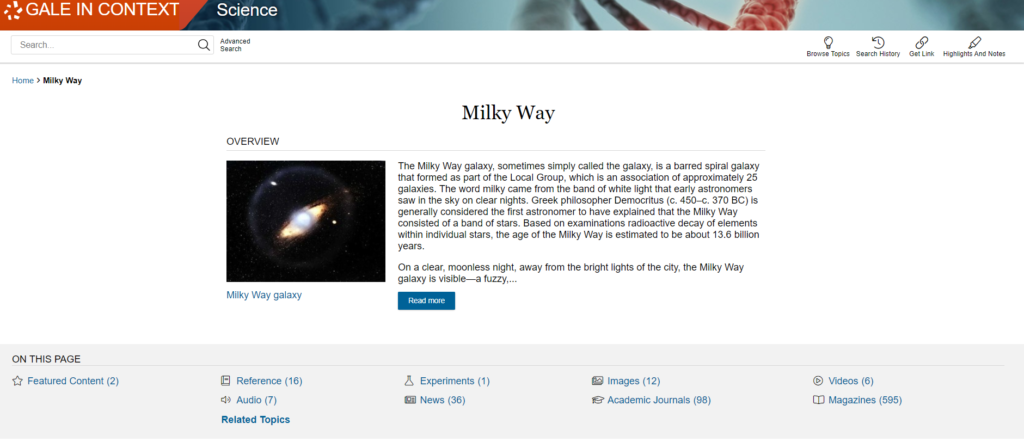
Lithium-ion batteries revolutionized portable computing, and these lightweight yet powerful batteries now charge devices ranging from cell phones to electric vehicles. Because lithium batteries depend on a recycling and renewable flow of electrons from anode to cathode rather than on the breakdown of chemicals, the batteries can also be charged and recharged, making them ideal for the storage of renewable solar and wind power. Indeed, as the Academy noted, lithium-ion batteries may make a “fossil fuel‒free society” possible.
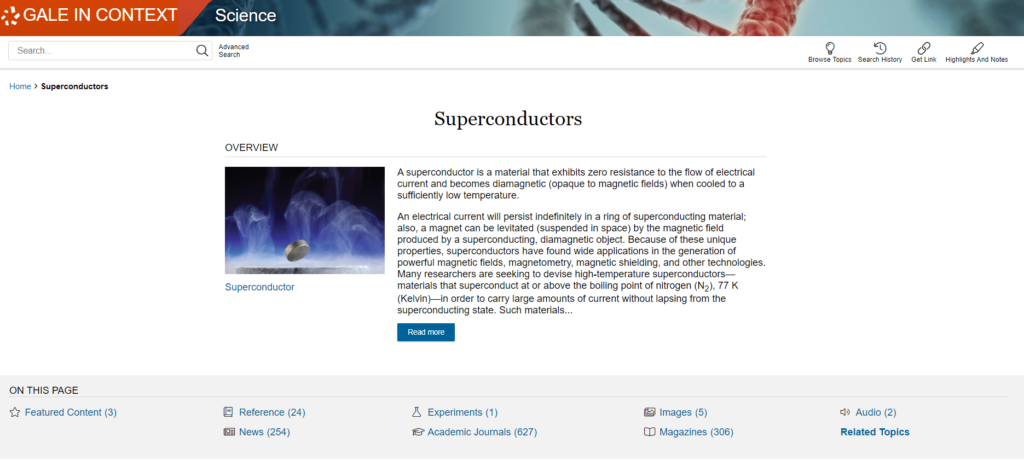
Whittingham’s work on superconductors in the 1970s helped identify an energy-rich material called titanium disulphide (which, at the molecular level, incorporates lithium ions) that he used to create a cathode. For an electron-emitting anode, Whittingham used metallic lithium to create a battery with high voltage potential. Further developments would help overcome the explosive nature of metallic lithium that made this early prototype battery unsuitable for general use. Goodenough subsequently used a cobalt oxide with intercalated lithium ions to create a cathode that nearly doubled the voltage potential of the lithium battery. In 1985, Yoshino’s work in Japan resulted in the creation of the first commercially viable lithium-ion battery that reduced the explosive dangers of earlier batteries by substituting carbon-based petroleum coke to intercalate lithium ions in the cathode. Starting in 1991, lithium batteries entered the general marketplace.
“For their discoveries of how cells sense and adapt to oxygen availability,” the Academy awarded the 2019 Nobel Prize in Physiology or Medicine jointly to American physician William G. Kaelin Jr. (1957‒), a professor at Harvard Medical School in Boston, Massachusetts, and investigator at the Howard Hughes Medical Institute; British-born scientist Sir Peter J. Ratcliffe (1954‒), a professor at Oxford University and director of clinical research at Francis Crick Institute, London; and American physician-scientist Gregg L. Semenza (1956‒), who, after graduating from Harvard, went on to study medicine at the University of Pennsylvania School of Medicine in Philadelphia. Semenza is now a professor at Johns Hopkins University and director of the vascular research program at the Johns Hopkins Institute for Cell Engineering.
As the Academy cited in a press release, “Oxygen sensing allows cells to adapt their metabolism to low oxygen levels: for example, in our muscles during intense exercise. Other examples of adaptive processes controlled by oxygen sensing include the generation of new blood vessels and the production of red blood cells. Our immune system and many other physiological functions are also fine-tuned by the O2-sensing machinery. Oxygen sensing has even been shown to be essential during fetal development for controlling normal blood vessel formation and placenta development.”
Understanding the cellular response to changing levels of oxygen is key to understanding multiple essential facets of cell physiology. The combined work of Kaelin, Ratcliffe, and Semenza articulated how—at the molecular and genetic level—cells both sense and adapt to changes in oxygen levels.
Semenza and Ratcliffe’s work focused on oxygen-dependent regulation of the EPO gene (initially in cells found in liver and kidney tissue, but ultimately across cells from multiple tissues) to help discover exactly how gene regulation is sensitive to oxygen availability. Kaelin’s work centered on research related to von Hippel-Lindau disease (VHL disease), an inherited syndrome that increases the risk of certain cancers in genomes with VHL gene mutations. Kaelin’s work with cancer cells showed that VHL was important in stimulating cell responses to hypoxia; Ratcliffe then demonstrated that VHL interacts physically with other known factors (e.g., HIF-1α) that are part of a cell’s sensing and response mechanism to changing levels of cellular oxygen.
The combined work of Kaelin, Ratcliffe, and Semenza also offers new insights on the pathology of disease as well as hope for new advances toward cures for anemias, cancers, and other diseases.
More biographical information to come in Gale in Context: Science and Gale in Context: Biography. Stay tuned!
Meet the Author
K. Lee Lerner is a writer, editor, and aviator who, along with Brenda Wilmoth Lerner, is the editor of the Gale Encyclopedia of Espionage, Intelligence, and Security, Climate Change in Context, and many other award-winning books and articles on science, technology, and a range of global issues. A full bio and list of his work may be found at https://scholar.harvard.edu/kleelerner and https://harvard.academia.edu/KLeeLerner/.


